Annotation
Ceramic and metallic designs do the same core job, but they do not behave in exactly the same way on the road. If you want a clear answer on the difference between a catalytic converter made from ceramic and one made from metal, the short version is simple: ceramic is common and cost-effective in high-volume production, while metallic designs can heat up faster, handle demanding layouts better, and support strong gas flow in compact spaces. That matters for drivers, repair decisions, and responsible recycling. Both types help reduce harmful exhaust emissions, yet they do not behave in exactly the same way. Material choice affects heat-up speed, airflow, durability, packaging, and, in some cases, long-term reliability.Key Takeaways
- Ceramic and metallic substrates do the same core job in different ways.
- Ceramic designs are common because they work well in large-scale vehicle production.
- Metallic designs can heat up faster and fit tighter exhaust layouts.
- Thermal durability is often a stronger point for metallic units.
- Precious metal coatings remain central in both designs.
- Replacement and recycling decisions should be based on the exact unit, not guesses.
Why the material inside the converter matters
Most drivers only notice this part when a warning light appears, performance drops, or a replacement becomes necessary. However, the internal structure plays a major role in how efficiently exhaust gases are treated. In simple terms, the substrate gives gases a path through the unit while exposing them to a coated surface where chemical reactions take place. That is the practical answer to what’s the purpose of a catalytic converter.
The original article already explains the problem clearly, and that point remains important because the function has not changed. Every vehicle with an internal combustion engine pollutes the air, and it doesn’t even matter how old it is. Some cars, especially diesel ones, produce more gasses than others. So that’s the problem, which requires a solution. That’s why every single one of them has a catalytic converter. There are wide varieties of them, but some are made of different materials than others.
That broad point is supported by the US EPA, which explains that transportation remains a major source of air pollution. So, when people ask what this part is really there for, the answer is direct: it helps reduce harmful emissions before they leave the tailpipe.
How ceramic converters are built
Ceramic units are widely used across the automotive industry. They usually contain a honeycomb substrate with many small passageways. This shape helps balance surface area with gas flow. As the exhaust passes through the channels, it comes into contact with the catalyst coating. That is how what catalytic converters do becomes more than a simple definition – it becomes a real process happening every time the engine runs.
The original article describes the standard setup in a way that still fits naturally here. In every diesel or petrol-powered car, a catalytic converter is installed right after the engine and into the exhaust system. It is made from metal or ceramic and shaped like a cylinder, with small holes for airflow. Without it, all the gasses that engines produce would reach our atmosphere. Some of them are hazardous to the environment and our health. Most used ceramic converters are made of cordierite formed into a honeycomb shape and have square or triangular passageways. It is coated with aluminum oxide or even more expensive ones (palladium, rhodium, or platinum).
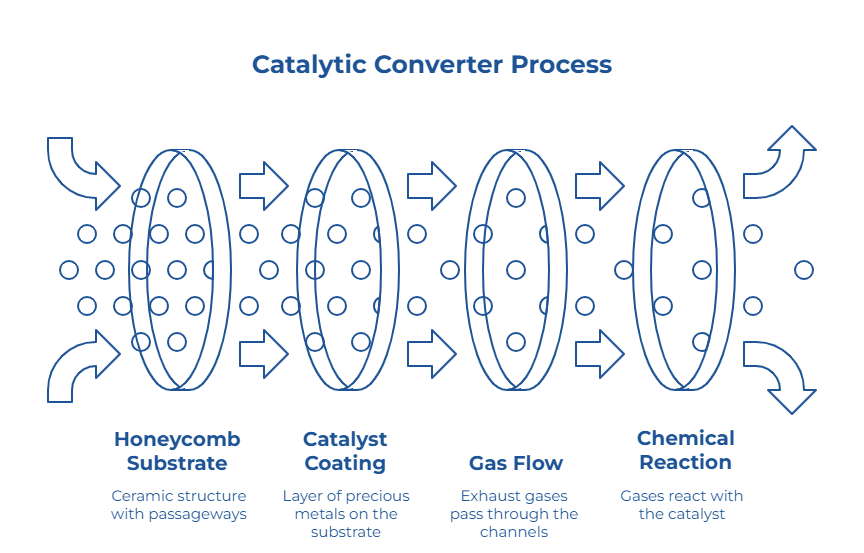
That description lines up with general industry information on ceramic substrates. According to Corning, ceramic honeycomb substrates are designed to provide a large active surface area while still allowing exhaust gases to pass through efficiently.
What makes metallic designs different?
Metallic substrates use thin metal foil formed into channels rather than a ceramic honeycomb block. The goal is the same, but the behaviour can differ. Metallic designs are often valued for quicker heat-up and greater flexibility in shape. That is why they are sometimes chosen for compact systems or demanding applications where exhaust packaging is tight and rapid operating temperature matters.
The older version of the article captures that contrast well, and it belongs within the same explanation rather than outside it. The difference between them is their price, difficulty in production, and efficiency. Most catalytic converters were made of ceramics. Still, nowadays, metallic ones are used more commonly. Mainly because they can offer a slightly smaller airflow resistance and start doing their job in lower temperatures. The gasses must reach a certain heat point for the catalytic converter to begin the cleaning process. Since the more minor airflow resistance is achieved with metal roll, it can provide way more surface for gasses to clean, compared to ceramic one.
Which one heats up faster?
Usually, metallic designs. That matters because the converter becomes more effective once it reaches the right temperature. Faster heat-up can mean earlier emissions control after start-up, which is useful in real driving conditions.
This point also appears in the original wording and still adds value when read in context. One significant advantage of metallic ones is more significant thermal conductivity. By that, we mean it is easier for this catalytic converter to heat up. And it can be placed closer to the car engine, where gasses are way hotter, and it can do its job in higher temperatures. When you think of other ones, the ceramic substance is very vulnerable to heat and might crack easily. Another metallic substance advantage is that it is easier to produce in different shapes. Hence, there is a possibility of flowing gas turbulence increasing. That means exhaust will use more catalyst area and convert even more emissions.
Comparison table: ceramic vs metallic substrate features
The table below shows the most practical differences between ceramic and metallic converter designs for everyday understanding.
| Feature | Ceramic substrate | Metallic substrate |
|---|---|---|
| Common use | Very common in mass-produced vehicles | Often used where packaging or heat-up speed matters more |
| Heat response | Effective, but often slower to warm | Usually warms faster |
| Shape flexibility | More limited | Greater freedom in design |
| Thermal stress | Can be more vulnerable to cracking | Often stronger under thermal shock |
| Large-batch production | Common and cost-effective | Can be more complex depending on the design |
Is metallic always the better choice?
No. For many standard road cars, ceramic remains a reliable and sensible option. It is proven, widely used, and practical in large production volumes. Metallic can offer gains in specific cases, but that does not make it the universal winner. The right choice depends on the engine, emissions system, and packaging needs of the vehicle.
The original text makes that point from a production angle, and it still deserves a place in the article. Many may think a metallic catalytic converter is a cheaper option, but that is not entirely true. First of all, the catalyst is made of a precious metal. Platinum itself is the best for converting processes and is used commonly. It is unsuitable in many ways, primarily because of unwanted chemical reactions and high costs. But it can do both rhodium and palladium functions (reduction and oxidation catalyst). Of course, nickel can do this process, but it is not legal in the European Union. And there is copper, which is illegal in North America. So, there is no way to make one catalytic converter that could be used worldwide.
If there is a need to make one metallic converter, it’s ok, but if we need a batch of them, then ceramic is the way to go. Mainly because it is easier and cheaper to make it in large quantities.
What should drivers and recyclers pay attention to?
Look at the exact unit, not assumptions. A ceramic structure does not mean low importance, and a metallic structure does not automatically mean better value or better performance in every vehicle. Internal coating, condition, fit for the vehicle, and end-of-life handling all matter. That is also why reading related guidance, such as Understanding the Types of Automotive Catalytic Converters, can help build a fuller picture before replacement or recycling decisions are made.
It is also worth keeping the environmental side in view. Blancomet’s guide to the environmental impact of catalytic converters explains why proper recycling remains important even after a unit is no longer fit for use.
Why recycling still matters after the converter fails
Both ceramic and metallic catalytic converters can contain valuable precious metals. Once a unit has reached the end of its service life, responsible recycling helps recover those materials and reduces waste. That is one reason the wider industry conversation often goes beyond emissions control and into resource recovery as well.
This is where the wider theme of Driving Sustainability and Economy Forward fits naturally. The converter is not only an emissions-control part. It is also a component that can continue to hold value after use when processed correctly by a specialist recycler.
Summary
Ceramic and metallic designs perform the same basic emissions-control role, but they differ in heat-up speed, airflow characteristics, thermal durability, and manufacturing flexibility. Ceramic remains highly relevant because it suits large-scale production and many standard vehicle applications. Metallic has clear strengths where fast warm-up and tighter packaging are needed. For owners, the practical lesson is simple: understand the unit fitted to the vehicle, replace it correctly, and, when it reaches the end of its life, use a trusted catalytic converter recycling service that can handle the material responsibly.
Frequently Asked Questions
Are ceramic converters still common?
Yes. Ceramic converters remain very common because they are effective, proven, and well suited to large production runs.
Do metallic converters always last longer?
Not always. They can offer stronger resistance to thermal stress in some conditions, but service life still depends on vehicle use, engine condition, and the overall exhaust system.
Do both types use precious metals?
Yes. Both rely on catalyst coatings that can include platinum, palladium, and rhodium.
Can a damaged converter still be recycled?
Yes. Even when a converter is no longer usable on a vehicle, it may still contain recoverable materials that make proper recycling worthwhile.
Is the material the only thing that matters?
No. The design, coating, condition, and suitability for the vehicle are all important when assessing performance, replacement, or recycling.

