Annotation
If you have ever wondered what sits inside a catalytic converter, the answer is both simple and impressive. From the outside, it looks like a sealed metal part in the exhaust system. Inside, however, it contains a carefully designed structure built to handle heat, guide exhaust gases, and support chemical reactions that make those gases less harmful. Knowing what is inside helps drivers understand how the unit works, why it fails, why recycling matters, and why the catalytic converter price is often linked to the materials hidden within it.Key Takeaways
- A catalytic converter contains a metal shell, substrate, washcoat, and precious metals.
- The substrate is usually ceramic, but some units use metal.
- Platinum, palladium, and rhodium help convert harmful gases into safer ones.
- The honeycomb design increases surface area for better exhaust treatment.
- Damage, contamination, and overheating can reduce converter performance.
- Recycling matters because spent units still contain recoverable valuable materials.
What is inside a catalytic converter?
At its core, a catalytic converter is a tough outer housing wrapped around a highly engineered inner structure. The purpose of that structure is to expose hot exhaust gases to a coated surface where chemical reactions can take place quickly and reliably. That is why the part is compact on the outside but complex within.
The original article introduces that idea well, and it still belongs here because it answers the main search intent clearly. Catalytic converters are indispensable components of modern vehicles, designed to reduce harmful emissions produced by internal combustion engines. These devices play a vital role in environmental conservation by converting dangerous pollutants into less harmful substances. But what exactly is inside a catalytic converter that enables this transformation? We will delve into the inner workings of catalytic converters to show their components and explain how they operate.
According to Britannica, these devices reduce harmful exhaust emissions through catalytic chemical reactions rather than by simply filtering gases. That point matters because many drivers assume the unit works like a basic trap, when in fact it works more like a reaction chamber.
The outer shell: why the casing matters
The external part is the first line of protection. The older version of the article describes it accurately, and it fits naturally into this section. The external part of the catalytic converter is a shell made of stainless steel, designed to endure high temperatures and protect the inner components. This casing serves as a robust container that withstands the heat and pressure generated by the vehicle’s exhaust system.
This casing does more than hold the internal parts together. It also protects the converter from road debris, moisture, vibration, and sharp temperature changes. Without a strong outer shell, the internal core could crack, loosen, or fail much sooner.
What sits at the centre?
The heart of the unit is the substrate. This is the core structure through which the exhaust gases pass. In many vehicles, it is ceramic. In some others, it is metallic. The shape is usually a honeycomb because that design gives a large contact area without blocking exhaust flow too much.
The original explanation is still useful here. Inside the casing lies the heart of the catalytic converter: the catalytic substrate. This is usually made of a ceramic material formed into a honeycomb structure, although some modern converters use a metallic substrate. The honeycomb design provides a large surface area, enabling maximum contact between the exhaust gases and the catalyst.
That honeycomb structure is not just clever design. It is essential to performance. According to Corning, ceramic honeycomb substrates are designed to provide high surface area while still allowing gases to move through efficiently. In simple terms, more surface area means more room for the chemical reactions that clean the exhaust.
Why is the washcoat important?
It spreads the active materials over a much larger surface.
The washcoat sits on top of the substrate and creates a rough, high-area layer that helps the precious metals do their job better. Without it, the active metals would have far less usable area. The article’s original wording explains this point well. Before the precious metals are applied, a washcoat is usually laid on the substrate. The washcoat, often made of materials like aluminum oxide, silicon dioxide, or titanium dioxide, serves to disperse the catalyst materials evenly. This significantly increases the surface area available for the catalytic reaction, making the conversion process more efficient.
This layer is easy to overlook, but it is one of the reasons a small unit can process a large volume of exhaust gases so effectively.
The precious metals inside
Many people ask what makes a catalytic converter special compared with other exhaust parts. The answer is the precious metal coating. The original article states it clearly: The Precious Metals: Platinum, Palladium, and Rhodium. The real magic happens due to the precious metals that act as catalysts: platinum, palladium, and rhodium. These metals are thinly coated on the substrate and are the agents that facilitate the chemical reactions to convert harmful pollutants into safer compounds. Platinum and palladium are primarily used for converting carbon monoxide and hydrocarbons into carbon dioxide and water. Rhodium is especially effective at converting nitrogen oxides into nitrogen and oxygen.
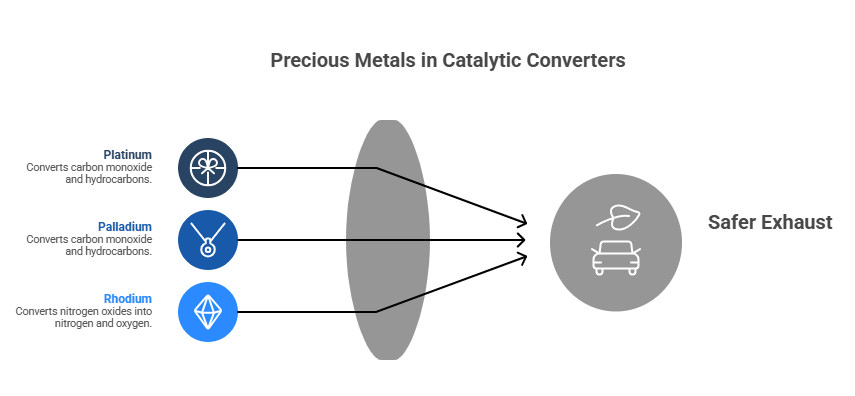
These metals are present in small amounts, but they are critical. They help speed up reactions without being consumed in the same way as fuel or oil. That is why an old, damaged, or used converter can still matter from a recycling point of view.
How do the chemical reactions work?
Hot exhaust gases pass through the honeycomb channels and touch the metal-coated surface. That contact helps convert harmful gases into less harmful ones.
The original article explains the process in a clear way that should remain part of the improved version. The Chemical Reactions. The basic idea is to facilitate a series of reduction and oxidation reactions. When exhaust gases pass through the catalytic converter, the catalysts trigger chemical reactions that transform harmful pollutants. Carbon monoxide is converted to carbon dioxide, hydrocarbons turn into water and carbon dioxide, and nitrogen oxides are reduced to nitrogen and oxygen.
That is the core function. It is not just about trapping pollution. It is about changing the chemistry of the exhaust before it leaves the vehicle.
Comparison table: what each internal layer does
The table below shows the main parts inside a catalytic converter and what each one contributes to the overall process.
| Internal part | What it is | Why it matters |
|---|---|---|
| Outer shell | Stainless steel casing | Protects the internal structure from heat, pressure, and impact |
| Substrate | Ceramic or metallic honeycomb core | Gives exhaust gases a large contact area |
| Washcoat | Surface layer applied to the substrate | Helps spread catalyst materials over more area |
| Platinum | Precious metal catalyst | Helps convert harmful exhaust compounds |
| Palladium | Precious metal catalyst | Supports oxidation reactions in exhaust treatment |
| Rhodium | Precious metal catalyst | Helps reduce nitrogen oxides |
Can the inside of a converter wear out?
Yes. Heat damage, contamination, melting, cracking, and physical impact can all reduce performance.
Even though the unit is built to last, it is not indestructible. The article’s existing section on recycling also explains why failure happens over time. Catalytic converters have a lifespan, often mirroring that of the vehicle itself, but they can degrade over time due to contaminants, melting, or mechanical damage.
That is why related guidance such as when you should replace the catalytic converter can help owners spot the signs before problems get worse.
Why does the inside affect recycling?
Because the value of a used catalytic converter is tied to the recoverable materials inside, not just the casing.
The existing text already makes this point and should stay in the article naturally. When they reach the end of their functional life, catalytic converter recycling is crucial. Not only do they contain precious metals that can be recovered and reused, but improper disposal can lead to environmental hazards. Additionally, the demand for the rare metals like platinum, palladium, and rhodium is ever-increasing, making their recycling both economically and environmentally beneficial.
This is also where searchers often become interested in the catalytic converter price. In practical terms, people are often asking about the value of the unit once it has been removed or reached end of life. While exact values depend on the specific converter and its internal content, the presence of recoverable precious metals is a key reason used units are collected and processed rather than thrown away. For more detail on how values are discussed, Blancomet’s article on catalytic converter values and vehicle examples provides relevant context.
What should vehicle owners remember?
Think beyond the metal shell. The important parts are hidden inside.
A converter may look like a simple exhaust component, but the inner design is what makes it effective. The substrate, washcoat, and precious metals all work together. That is why proper diagnosis matters when a unit fails, and it is also why correct disposal matters once it is no longer usable.
The older conclusion still works well once it is placed in the wider context of the updated article. The catalytic converter is a marvel of chemical engineering that plays a critical role in reducing vehicle emissions. With its stainless steel casing, catalytic substrate with a honeycomb structure, washcoat for dispersion, and precious metal catalysts like platinum, palladium, and rhodium, it performs a series of complex chemical reactions seamlessly and efficiently. Understanding its components and how they work can give us a greater appreciation of this essential device that aids in the fight against air pollution. Just do not forget that catalytic converter recycling is also needed.
Summary
Inside a catalytic converter, there is far more than an empty chamber. There is a stainless steel shell, a ceramic or metallic honeycomb substrate, a washcoat that expands surface area, and precious metals that drive the chemistry of emissions control. Together, these parts reduce harmful gases and make modern exhaust systems far cleaner than they would be otherwise. They also explain why the catalytic converter price is often linked to what remains inside a used unit. When that unit reaches the end of its life, responsible recycling through a specialist service helps recover useful materials and supports a cleaner process from start to finish.
Frequently Asked Questions
What is the main material inside a catalytic converter?
The main internal structure is usually a ceramic honeycomb substrate, although some converters use a metallic substrate instead.
Why are precious metals used inside catalytic converters?
Platinum, palladium, and rhodium help trigger the chemical reactions that convert harmful exhaust gases into less harmful substances.
Does every catalytic converter have the same internal design?
No. The overall layout is similar, but substrate type, coating, and internal construction can vary between vehicle models and converter types.
Can the inside of a catalytic converter get damaged?
Yes. Overheating, contamination, impact, or age can crack, melt, or weaken the internal substrate and reduce performance.
Why should a used catalytic converter be recycled?
Because even a failed unit may still contain recoverable precious metals, and correct recycling helps reduce waste and support resource recovery.

